Scientists have found catalysts that improve an important industrial reaction and make it more eco-friendly.
Without needing high temperatures or toxic catalysts, scientists at Nagoya University in Japan have developed a chemical reaction that produces high yields of a compound used in a wide variety of industries. The approach was described in the journal ACS Catalysis and offers a useful, sustainable solution for industrial (meth)acrylate ester synthesis.
(Meth)acrylate esters are used in industrial coatings and masonry and to make plastics, dyes and adhesives. But the chemical process for making (meth)acrylate esters from methyl (meth)acrylates involves high temperatures, long reaction times and toxic compounds. It also can result in unwanted side reactions.
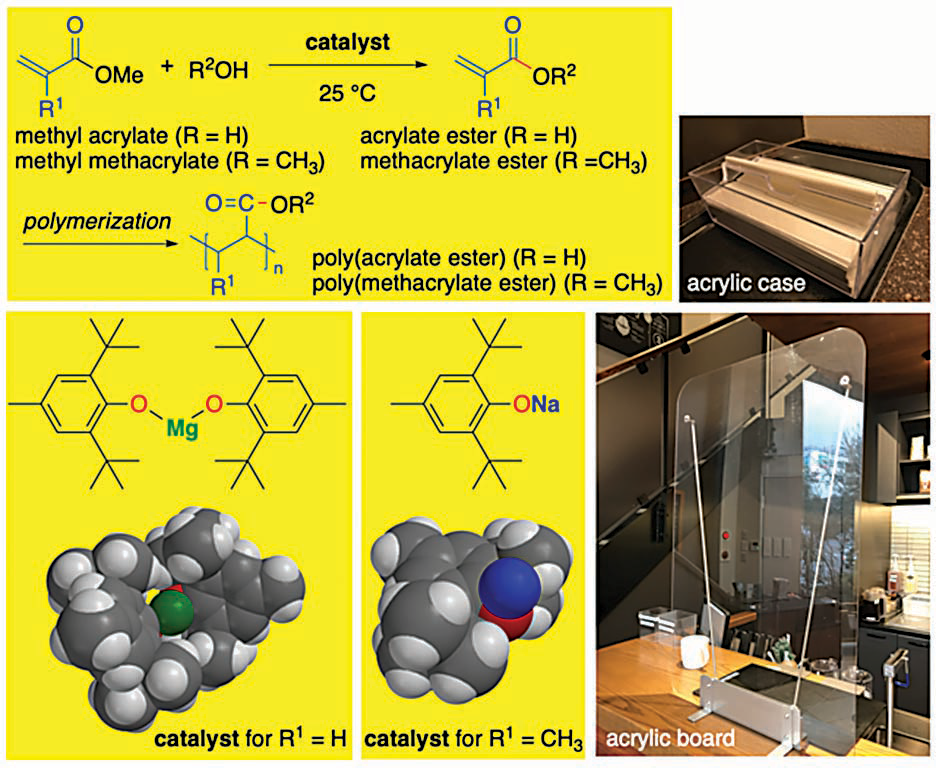
Sodium and magnesium aryloxides can catalyze the transesterification of methyl (meth)acrylates at room temperature with high chemoselectivity, producing high yields of (meth)acrylate esters without the use of toxic metals or ligands. Image courtesy of K. Ishihara
Scientists, including Nagoya University professor Kazuaki Ishihara and his colleagues, have been working on improving this process to make it more eco-friendly. They specifically worked on bettering the catalyst involved in the chemical reaction that turns methyl (meth)acrylates into (meth)acrylate esters, which is called transesterification.
“Millions of tons of (meth)acrylate esters are produced annually and are among the most important manufactured chemicals around,” he said. “Their transesterification, using alcohol and a catalyst, fine-tunes their properties, producing a wide range of (meth)acrylate esters.”
Ishihara and his colleagues found that sterically bulky sodium and magnesium aryloxides worked very well as nontoxic alternatives. They catalyzed the transesterification of methyl (meth)acrylates at the relatively mild temperature of 25 degrees C (77 F), producing high yields of a broad range of (meth)acrylate esters depending on the type of alcohol used in the reaction.
The team also conducted computational calculations to uncover the details of what happened during the chemical reaction, showing that it had high chemoselectivity. In other words, the reaction happened the way the scientists wanted — without undesirable side reactions.
“Our transesterification process is a practical and sustainable candidate for industrial (meth)acrylate ester synthesis,” Ishihara said, “providing excellent chemoselectivity, high yields, mild reaction conditions and a lack of any toxic metal salts.”
The team next aims to collaborate with colleagues in industry to use the approach in (meth)acrylate ester production. The scientists also intend to continue searching for efficient catalysts for the transesterification of methyl (meth)acrylates and to develop recyclable catalysts.
— Nagoya University

