
Courtesy of All images: Miraclean
When cleaning parts, there is general cleaning and then there is precision cleaning. Both types are important in medical device manufacturing, but precision cleaning—repeatable removal of contaminants to a specified, usually validated standard—has become more prevalent in part because of increasingly stringent Food and Drug Administration oversight of the manufacturing process, resulting in stricter process QC.
Precision cleaning takes performance up a few notches over general parts cleaning. It is typically based on a written cleaning specification to meet company and customer quality goals and FDA requirements. Having a validated, written specification and demonstrating that the user abides by it repeatedly (repeatability) are important to FDA compliance. To ensure repeatability, precision cleaning systems are usually automated.
Precision Cleaning Stations
There may be one or more cleaning stations, or tanks, in a precision cleaning line for medical parts and implants. These may be equipped identically, or they may differ depending on the cleaning goals, variety of parts being processed, soil loading and types of cleaning chemistries required. Precision cleaning tanks tend to be equipped with multiple types of mechanical actions to enhance performance.
Because ultrasonics offer gentle, effective cleaning for a variety of part geometries and substrates, they are often specified for precision cleaning tanks. They are available in different frequencies appropriate to the substrates being cleaned and the cleaning requirements. The microscopic bubbles created by ultrasonic energy travel throughout the bath and into part apertures, no matter how complex. Their implosion creates a gentle but thorough cleaning action that breaks soil bonds. The lower the frequency, the larger the bubbles produced by the sound waves and the more aggressive the cleaning action.
For example, a frequency of 25 kHz is more aggressive than 40 kHz, with 40 kHz being the universal, or most commonly applied, frequency. Ultrasonics are also available in higher frequencies, such as 68 kHz and above, as well as multiple frequency units, which may be suitable for delicate parts and micropart geometries.
In addition to ultrasonics, other potential mechanical cleaning actions include vertical agitation, spray, spray under immersion and rotation. The selected combination of methods depends on part geometry, substrate, soil type and cleaning chemistry. Racking or fixturing of the parts to optimize impingement from these mechanical actions is also important. This includes fixturing so that spray makes contact where appropriate. Custom fixtures and racks can be designed to maximize exposure to spray, ultrasonics and agitation.
Automated Control
A precision ultrasonic cleaning tank typically incorporates three other features: heat to optimize chemistry performance and, thus, cleaning effectiveness; a filtration loop to remove particulates and extend bath life; and a surface sparge to skim floating soils to a separate chamber, or weir, prior to the removal of cleaned parts. A fourth feature that may be important is automatic chemistry control, which can be enabled by conductivity monitoring and automatic replenishment. This allows a system to continuously operate within the validated range (instead of periodic, manual control).
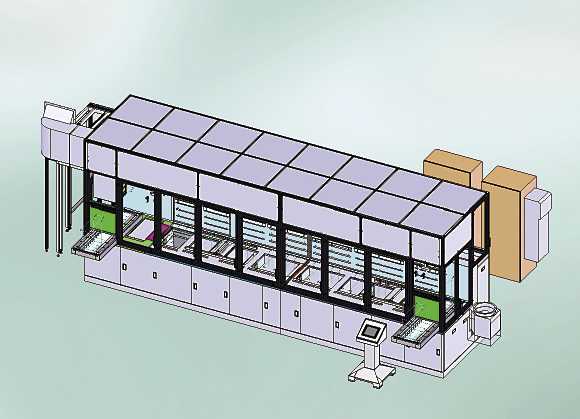
An automated precision cleaning and passivating line with system enclosure.
In an automated precision cleaning line, all of these features are controlled by programmable automation. The automation also advances each process load through each step in the program selected for that load. Most systems come with multiple automated programs. It is possible to vary the stations that a particular program includes, as well as station dwell times. Programs may be modified on the shop floor using password-protected screens. There may also be a telephone dial-in feature to make modifications or troubleshoot problems as needed.
To meet exacting verification requirements for medical parts and implants, critical process parameters, such as time, temperature and chemistry concentration, can be continuously monitored by system automation. The readings register in real time on the operator, or human/machine, interface. This interface is typically a color touch screen. Critical process data associated with these readings can be captured and stored electronically or printed as required to meet the FDA’s CFR 21, part 11 requirements. Conditions out of the allowable range can be set to alarm and register on the HMI.
Thorough Rinsing
Cleaning is followed by thorough rinsing. On a precision cleaning line, this typically means a series of two or more rinses after each different chemistry station on the line. These may be set to counterflow, which means freshwater feed is the second rinse, which backflows to feed the first. The same water is then used twice to reduce consumption while maximizing rinse performance.

An ultrasonic cleaning tank configuration with spray-off capability and a four-sided weir is common when cleaning medical parts.
Precision rinse tanks are typically heated and may be filtered to a process-appropriate micron level, particularly in later-stage manufacturing operations, clean room applications or whenever part geometries or cleanliness standards warrant. Some precision rinse tanks may be equipped with ultrasonics, agitation or other mechanical actions, depending on part complexity.
When spot-free rinsing is required, either deionized or reverse-osmosis water is the recommended feed on the precision cleaning line. Rinses may be continuously flowed or flowed in combination with rinse QC, which measures allowable levels of conductivity or resistivity in the rinse water and calls for a fresh feed of water as needed. Critical parameters for rinsing, including flow and conductivity/resistivity levels, may be continuously monitored by system automation and recorded for conformance verification.
Nitric or Citric Passivation
Passivation is typically required in precision parts cleaning. It can be implemented in a batch-style, immersion precision cleaning line.
Passivation removes free iron from part surfaces. Passivation of medical devices and implants typically conforms to ASTM specification A967—which provides a number of possible recipes based on temperature, concentration and time, as well as testing protocols to verify passivation success. The A967 specification allows for either nitric or citric passivation. Some manufacturers are shifting from nitric to citric, as it is more operator- and environment-friendly.
Passivation takes place in an immersion tank and becomes another step in a multiple-station precision cleaning process. As with the other stations on the line, critical process parameters for the passivation tank can be captured and recorded to verify conformance. Ideally, passivation is followed by two or more counterflow rinses.
Cleaned and rinsed parts may be dried in a recirculating hot-air dryer. In higher-throughput operations, a second dryer may be added to decrease cycle time. Another variation is a drying tunnel. When cleaning success is measured by particle counts, the dryer may be equipped with a HEPA filter to remove particulate. This option is typically used in clean room applications.
Sizing the System
The configuration and size of any parts cleaning system depends on several key items. The tank work area is determined by the largest possible batch size. The number of cleaning stations in a line depends on the level of soil loading, the variety of substrates to be processed and the process goals, including the optional use of passivation. The number of rinse stations depends on the number of cleaning stations. The number of drying stations depends on throughput goals for the precision cleaning line.

The cleaning end of a precision cleaning and passivating line for medical devices.

Interior work zone of a small-footprint, precision cleaning system featuring ultrasonic cleaning, ultrasonic rinsing and air blow-off.
Ultrasonic parts cleaning systems can be quite large, but they don’t have to be. A new breed of ultrasonic machine delivers a more cellular approach in a smaller footprint. This type of system features precision ultrasonic cleaning and rinsing and high-powered air blow-off in a fully enclosed unit, such as Miraclean’s SonicCell Cube, which measures 72 "×60 ". The operation is controlled by a programmable logic control and critical process data can be captured and electronically stored or printed, as with larger systems. This type of system may be a good starting point for many manufacturers implementing precision cleaning because of its small size and lower cost.
The need for equipment validation is one of the biggest changes in medical device cleaning. New precision cleaning equipment that has been specified, ordered and delivered typically must be validated through tests designed by the end user to demonstrate the system’s ability to produce acceptable parts. Cleaned sample parts are tested to demonstrate conformance to the specification. Once validated, the equipment is expected to produce product within the ranges proven in the validation testing.
Whatever the projected scope of the equipment, test cleaning parts is critical when defining a new cleaning specification or when implementing an existing cleaning specification in new equipment for cleaning medical parts and implants. Be sure the proposed system has enough capability and versatility to successfully and repeatably clean production-size loads. This may increase the initial system price, but, over the life of the equipment, it can mean the difference between accepted and rejected parts. CTE
Related Glossary Terms
- feed
feed
Rate of change of position of the tool as a whole, relative to the workpiece while cutting.
- micron
micron
Measure of length that is equal to one-millionth of a meter.
- ultrasonic cleaning
ultrasonic cleaning
Method of cleaning metal or plastic parts by immersing them in an aqueous or solvent-based cleaning solution and imposing ultrasound energy on the bath to enhance cleaning by creating cavitation conditions at the part surface, which imparts a strong scouring action to remove tenacious soils.

